ALLEVYN COMPLETE CARE Dressing mode of action
Chronic wounds can cost you

Patients with HAPIs in the US: Cost an additional $21,767 and spend an extra 9.5 days in hospital5

1 out of 10 patients are affected by hospital-acquired pressure injuries (HAPIs)6
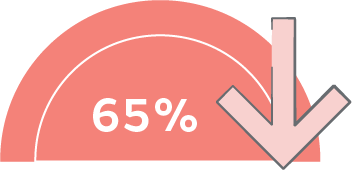
ALLEVYN 5 Layer Dressings reduce pressure injuries by more than 65% and associated costs with ShearDEFENSE◊ Technology***7-9
Reasons to change to ALLEVYN COMPLETE CARE Foam Dressings


Multi-Layer Design
Our unique, 5-layer construction absorbs fluids and shear forces, and redistributes pressure.*10-14
*As demonstrated in vitro
Various fits
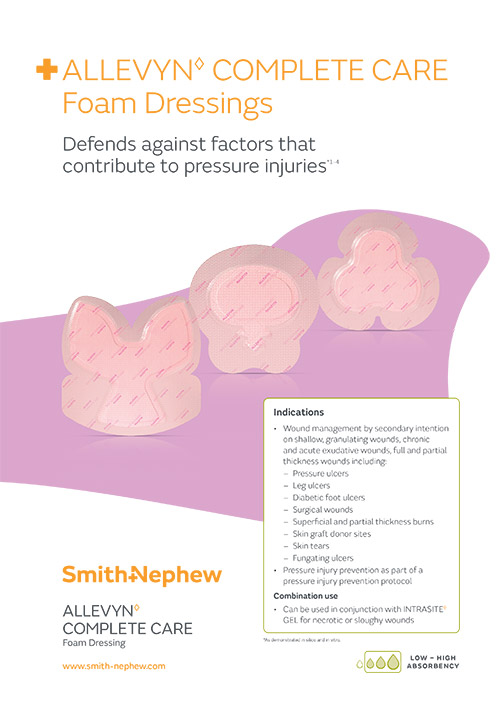
ALLEVYN COMPLETE CARE Dressings can be used to help prevent medical device-related pressure injuries*, dressings come in multiple shapes and sizes to securely fit body contours and help ensure the dressing stays in place. 2-4,7,26,30
*as part of a prevention bundle Refer to IFU – MDRPIP is covered under PIP.
Pressure injury prevention
ALLEVYN 5 Layer Dressings reduce pressure injuries by more than 65% and associated costs.***3-5
***Reduction of per-patient cost of 37-69%; compared with using standard preventive care alone; n=359
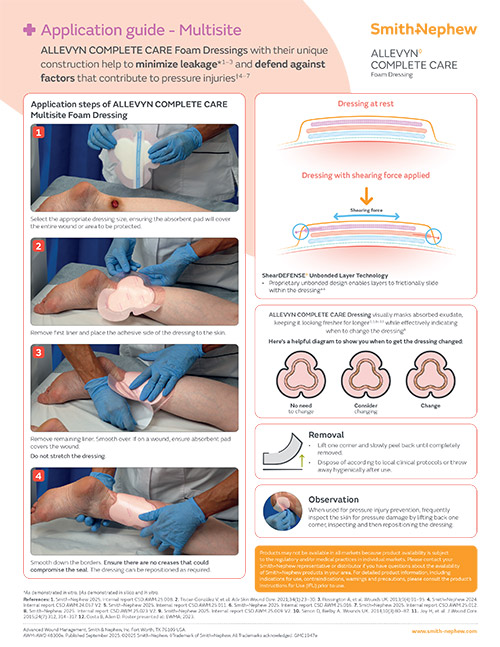
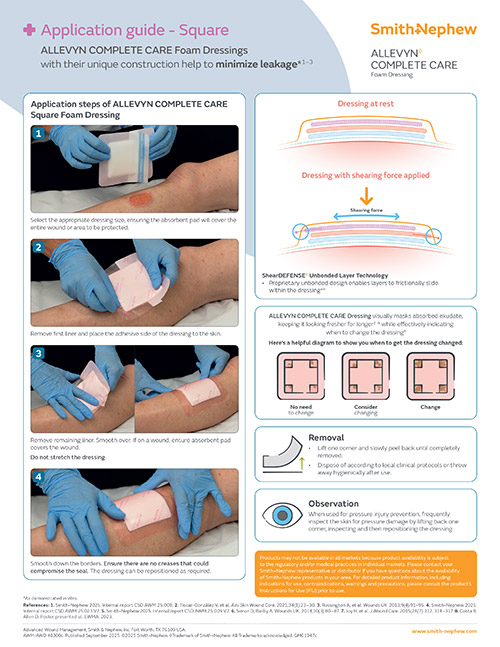
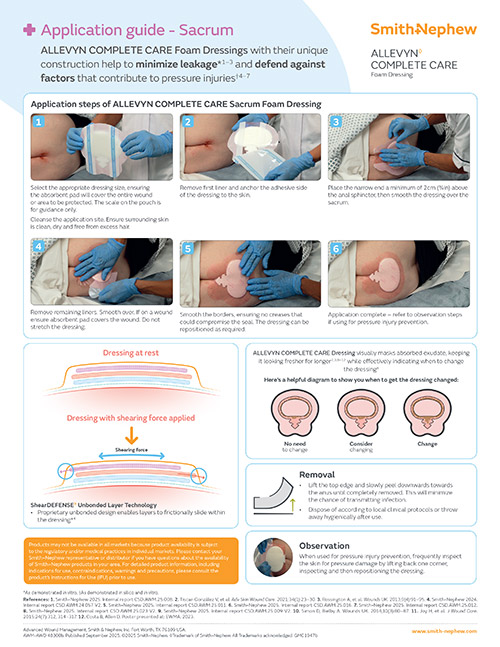
Visual indicator
The EXUMASK◊ Change Indicator serves as a dressing change indicator, helping avoid clinically unnecessary dressing changes, freeing up time and making life simpler.26,27
*Based on ALLEVYN 5-layer Dressing evidence
Longer lasting
ALLEVYN 5 Layer Dressings can be worn nearly 30% longer than other foam dressings*7 and remains in place for up to 7 days.§20
*versus previous dressings (includes: other foam dressings, gelling fibers, protease-modulating dressing, contact layers, etc.); Mean difference of -0.85 change per week (2.91 vs. 2.06); 95% CI: -1.62 to -0.09; p=0.029; n=307
§ As demonstrated in a volunteer trial. 120 volunteers wore both dressings on intact skin for up to 7 days on their thighs or shins
ShearDEFENSE Technology
ALLEVYN COMPLETE CARE Dressing is a unique dressing that has been proven to deliver frictional energy absorber effectiveness within the dressing due to its truly independent layers,**13, reducing soft tissue strain.**13,28,29
**As demonstrated in vitro
New evidence
ALLEVYN COMPLETE CARE Foam Dressings absorb up to 93% of mechanical energy into its internal layers15
*FEAE evaluates how well a dressing with able-to-slide layers can absorb mechanical energy that contributes to pressure injuries.
How it works
- Breathable21 bacterial barrier22
- EXUMASK◊ visual indicator
- EXULOCK◊ lock-in technology
- Absorbent hydrocellular foam
- ComfortSTAY◊ gentle silicone adhesive11,23-25
Patient support material
Connect with us to request a demo
Share your email address for more information about how ALLEVYN Dressings can help your facility reduce costs and patient recovery times.

Related products
* As demonstrated in Finite Element Modelling (FEM) model of the heel. ALLEVYN COMPLETE CARE and Mepilex Border Dressing as prophylactic dressings used to protect the heel soft tissue. Internal report: CSD.AWM.25.024
**performance relates to soft tissue strain
***Reduction of per-patient cost of 37-69%; Compared with using standard preventive care alone; n=359.
Ǿ As demonstrated in a volunteer trial. 120 volunteers wore the dressing on intact skin for up to 7 days on their thighs or shins.
- ALLEVYN COMPLETE CARE and Mepilex Border Dressing as prophylactic dressings used to protect the heel soft tissue. Internal report: CSD.AWM.25.024
- Smith+Nephew 2025. Conformability Testing of ALLEVYN COMPLETE CARE Dressing. Internal report: CSD.AWM.25.013.
- Smith+Nephew 2025. Extensibility Testing of ALLEVYN COMPLETE CARE Dressing. Internal report: CSD.AWM.25.012.
- Tiscar-González V, Menor-Rodríguez MJ, Rabadán-Sainz C, et al. Clinical and Economic Impact of Wound Care Using a Polyurethane Foam Multilayer Dressing. Adv Skin Wound Care. 2021;34(1):23-30.
- Wassel CL, Delhougne G, Gayle JA, Dreyfus J, Larson B. Risk of readmissions, mortality, and hospital-acquired conditions across hospital-acquired pressure injury (HAPI) stages in a US National Hospital Discharge database. Int Wound J. 2020;1–11. https://doi.org/10.1111/iwj.13482.
- Li Z, Lin F, Thalib L, Chaboyer W. Global prevalence and incidence of pressure injuries in hospitalised adult patients: A systematic review and meta-analysis. Int. J. of Nursing Studies. 2020;105:103546
- Atkinson, L. & Costa, B. (2024). “Pressure Injury Prevention with A Unique Multi-Layer Foam Dresing: A Systematic Review and Meta-Analysis of Randomized Controlled Trials” Poster presented at the 34th European Wound Management Association Annual Meeting, May 1-3, 2024, London, UK.
- Marché C, Creehan S, Gefen A. The frictional energy absorber effectiveness and its impact on the pressure ulcer prevention performance of multilayer dressings. Int Wound J. 2024 Apr;21(4):e14871. doi: 10.1111/iwj.14871. PMID: 38591160; PMCID: PMC11002638.
- Forni C, Searle R. A multilayer polyurethane foam dressing for pressure ulcer prevention in older hip fracture patients: an economic evaluation. J Wound Care. 2020 Feb 2;29(2):120-127. doi: 10.12968/jowc.2020.29.2.120. PMID: 32058851.
- Smith+Nephew 2025. Free swell Absorbency and Retention after Compression Testing of ALLEVYN COMPLETE CARE Dressing. Internal report: CSD.AWM.25.008.
- Rossington A, Drysdale, K, and Winter R. Clinical performance and positive impact on patient wellbeing of ALLEVYN Life. Wounds UK. 2013;9(4):91-95.
- Smith+Nephew 2025. Wound Models testing over 4 Days – Horizontal, Moderate Flow, Viscous SWF and Wound Models Testing over 7 Days, Horizontal, Moderate Flow of ALLEVYN COMPLETE CARE Dressing. Internal report: CSD.AWM.25.009 V2.
- Smith+Nephew 2024. Comparison of Frictional Energy Absorber Effectiveness (FEAE) in two five-layer hydrocellular polyurethane foam dressings (HPFD) containing superabsorber and masking layers. Internal report: CSD.AWM.24.057 V2.
- Smith+Nephew 2025. Pressure redistribution testing of the ALLEVYN COMPLETE CARE dressing. Internal report: CSD.AWM.25.016.
- Orlova D, Orlov A, Gefen A. The Protective Efficacy of a New Soft Silicone Multi-Layer Dressing in Reducing the Heel Pressure Ulcer Risk. International Wound Journal. 2025;22(10):e70764
- Austin M. Implementation of a Medical Device Related Pressure Injury Prevention Bundle: A Multidisciplinary Approach. Paper presented at: SAWC; 2019.
- Mcfee K. Implementation of medical-device related pressure injury prevention protocols: Prophylactic foam dressing packets for proned Covid-19 patients in the ICU. Paper presented at: NPIAP; 2022; Florida.
- Joy H, Bielby A, Searle R. A collaborative project to enhance efficiency through dressing change practice. J Wound Care. 2015;24(7):312, 314 – 317.
- Smith+Nephew 2025. Waterproofness testing of the ALLEVYN◊ COMPLETE CARE dressing. Internal report: CSD.AWM.25.017
- Smith+Nephew 2025. Topline results from ALLEVYN◊ COMPLETE CARE Foam Dressing volunteer trial (HVS2313) on thighs and shins. Internal report: CSD.AWM.25.032.
- Smith+Nephew 2025. Moisture Vapour Permeability Testing of ALLEVYN COMPLETE CARE Dressing. Internal report: CSD.AWM.25.011.
- Smith+Nephew 2025. Bacterial barrier testing of the ALLEVYN COMPLETE CARE dressing. Internal report: CSD.AWM.25.014
- Vowden K, Moiemen N, Drysdale K, Mistry C. An open, prospective randomised, multi-centre clinical evaluation of a hydrocellular silicone foam dressing* in the management of exuding chronic and acute wounds. Paper presented at: EWMA; 2011; Brussels.
- Hurd T, Gregory L, Jones A, Brown S. A multi-centre in-market evaluation of ALLEVYN◊ Gentle Border. Wounds UK. 2009;5(3):32 – 44.
- Smith+Nephew. Internal Report. EA_AWM_RENASYS_009_v1
- Costa B, Allen D. Results from an international survey: foam dressings with WTCI may enhance clinician confidence to extend wear times compared with other foam dressings. Poster presented at: the 33rd conference of the EWMA; May 3‒5, 2023; Milan, Italy.
- Hurd T, Allen D, Saunders C. Reduced weekly dressing changes with a five-layer foam dressing compared with other previously used dressings in wounds of mixed aetiology: results of a systematic literature review and meta‒analysis of clinical studies. Poster presented at: the 33rd conference of the EWMA; May 3‒5, 2023; Milan, Italy.
- Smith+Nephew 2024. Finite Element Modelling (FEM) of ALLEVYN COMPLETE CARE Dressing as a prophylactic dressing used to protect the posterior heel. Internal report: CSD.AWM.24.058 V2
- ALLEVYN COMPLETE CARE and Mepilex Border Dressing as prophylactic dressings used to protect the heel soft tissue. Internal report: CSD.AWM.25.024
- Stephen-Haynes J, Bielby A, Searle R. The clinical performance of a silicone foam in an NHS community trust. Journal of Community Nursing. 2013;27(5):50 – 59.
Recall and Complaints
Customer will provide such support and assistance as S+N may reasonably request in the event of a general or limited voluntary or mandatory recall of the Product(s). Customer shall promptly report any complaint in respect of the Products to complaints@smith-nephew.com.